Not long ago, it was admitted that springtails did not master their jumps to the point of making it an ordinary means of locomotion. These jumps, resulting from an explosive relaxation of their furca, were then considered as an escape reflex random trajectories. However, chapter 24, devoted to the collophore, shows that this organ can be used as a means of attachment to the substrate, when receiving a jump. We can therefore consider that the springtail has, at least partially, an ability to control certain phases of its flight.
The dynamics of animal locomotion is arousing growing interest among biologists, engineers and mathematicians who see innovative potential in it,both in sectors such as robotics and medical devices (prostheses). We know that the locomotion of arthropods represents one of their highest energy expenditures. Indeed, the energy balance of their locomotion deteriorates as much as their body mass decreases. During their evolution, springtails, whose weight is very low, were therefore probably led to adopt models likely to improve the cost/efficiency ratio of their locomotor system. The ability to flee from danger is, moreover, one of their main conditions for survival.
To approach the mechanisms at play during the jump, several components must be taken into account. For example, the nature of the impulse initiated by the furca or the movements and postures of the springtail's body during flight. The shots taken on jumps, as well as the in-depth morphological studies, bring new elements which were not accessible to conventional observation, if only because of the brevity of the events concerned, quantifiable in ms (1/1000 second) or the morphological complexity of the jumping apparatus. We are now able to measure speeds and accelerations during flights and to take into account, thanks to computer modeling, aerodynamic parameters (effects of air on moving bodies) or hydrodynamics (effects of liquids incompressible on moving bodies ).
I evoke here, "without equation", the approach of the scientists on the kinematics of the jumps of certain springtails. This chapter was inspired by " Directional takeoff, aerial recovery and landing in adherence of semi -aquatic springtails " (Nov. 2022) [1], publication of a team of researchers from the University of Maine (USA). I contacted one of the co-authors, Victor Ortega Gimenez, who sent me videos which he authorized me to use. For written transcription purposes, part of the illustrations which follow are therefore copies of images selected from video sequences. Finally, some of the diagrams that follow are adaptations made by me, because the aforementioned study is protected by copyrights*.
Elements of morphologies:
In addition to the chapters about collophorus, I mention here some determining elements of anatomy during jumps. Some of the following illustrations come from an article written by Fabio Goncalves de Lima Oliveira [2]: “ On springtails: a morpho-functional study of the jumping apparatus ”. Observation with the naked eye does not make it possible to apprehend the complexity of the springtail's jumping apparatus, long considered a simple catapult. Below, two positions of the furca (F) of an Entomobryomorph. The basal sclerite rods (BP1, 2, 3) are shown in color.
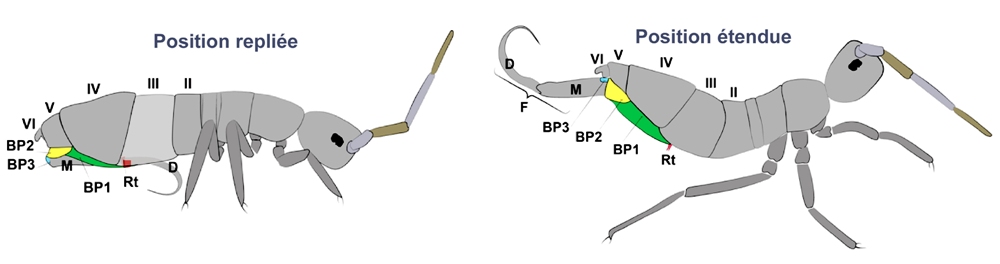
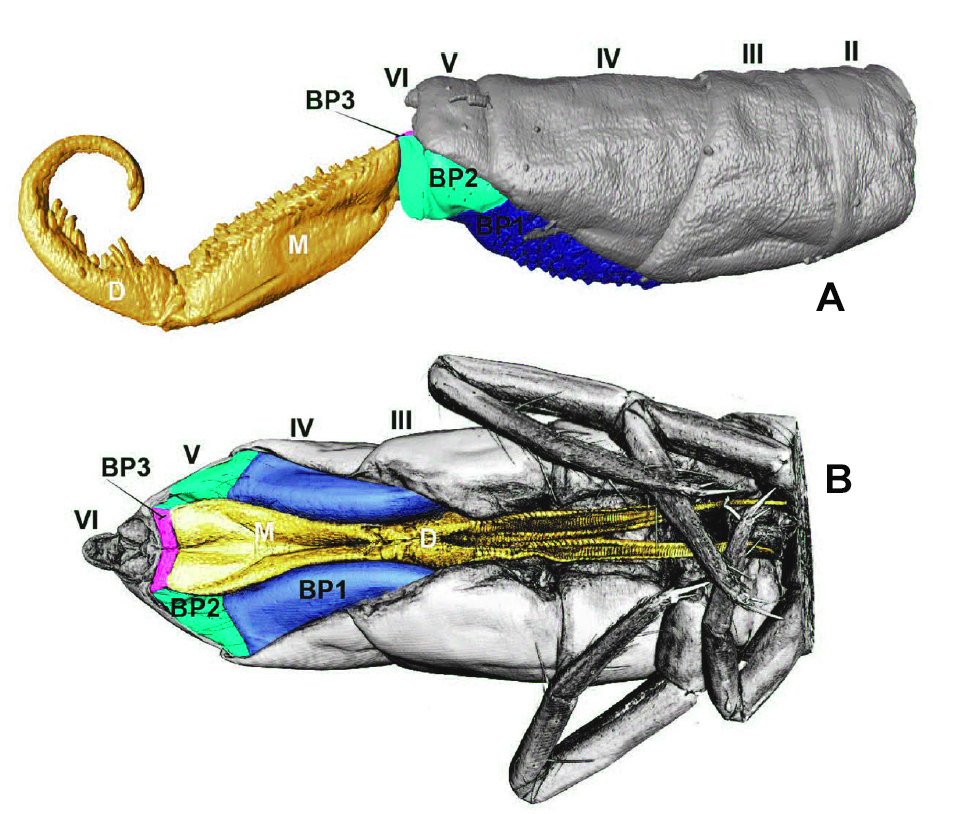
Below, MicroCT reconstruction (see Orchesella cincta basal plates page. Figure “A” is a lateral view, from the second abdominal segment to the end of the furca. It corresponds to the diagram on the previous page “extended position.” Figure “B” is a ventral view of the jumping device with the furca in the folded position, fixed by the retinaculum.
Scientists who wonder about the nature of the mechanisms that cause the jump put forward two hypotheses:
1) The elastic energy (resulting from muscular action) necessary for the spring mechanism would be directly stored in the basal sclerite of the fourth abdominal segment.
2) The jump is triggered by a brief increase in hemolymphatic pressure.
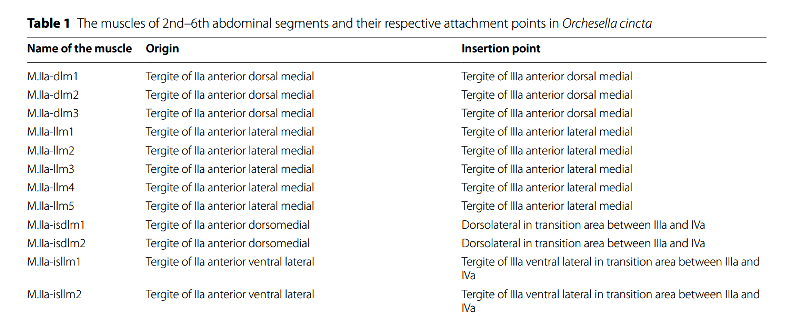
Studies to date suggest that the answer may be a combination of the two: “muscular system + hydraulic pressure”. In order to understand these morpho-functional aspects, the researchers have deepened the anatomical study of the jumping apparatus, as shown below by the microCT* morphological reconstruction in internal lateral view of abdominal segments 2 to 6. the muscles and cuticle of the jumping apparatus of an Orchesella cincta . Each muscle is listed and classified in a nomenclature which defines its name and the points of attachment of its two extremities (see extract below the illustration).
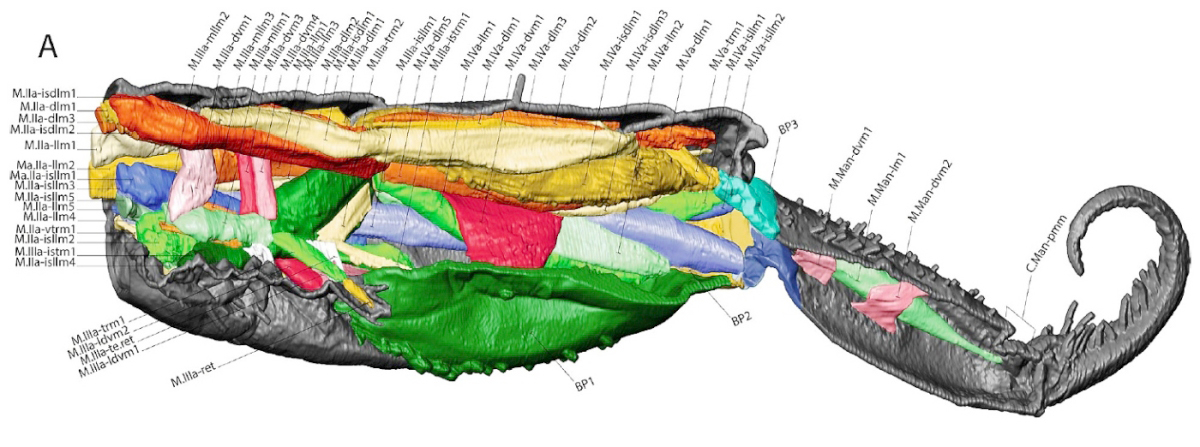
* Note : The level of detail, essential to address the mechanisms of muscle relaxation, is almost inaccessible by conventional methods, based on observation after dissection. It is only accessible by using the technology called "micro-CT" or micro-tomodensitometry. This reconstruction in 3D (three-dimensional) imaging uses X-rays to see inside a subject. It generates detailed images of the internal structure of small samples and allows resolutions of 100 nanometers, or 0.001 mm. This microscopic precision is obtained with an assembly like the one described below.
Methodology :
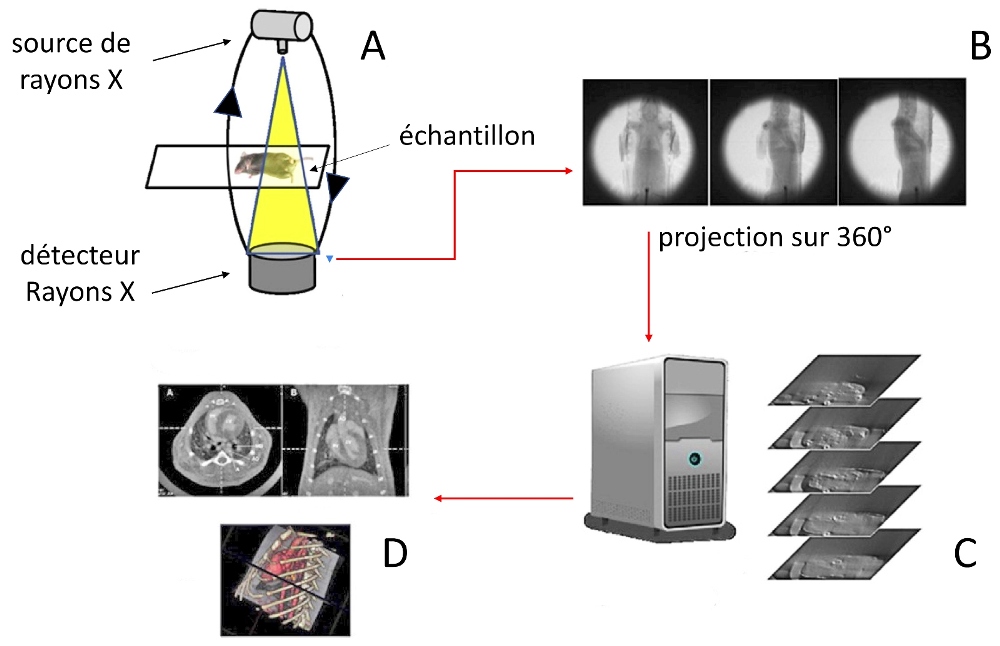 - The sample at to study is exposed to an X-ray source (A).
- The sample at to study is exposed to an X-ray source (A).
- An angular scan (by rotation) is performed around this subject to take, slice by slice, several hundred two-dimensional images (B).
- Computer pre-processing (C) of these shots (photometric calibration) transforms them into logarithms. Process also named: Cone-beam X-ray transforms of linear attenuation coefficients.
- These logarithms are processed by calculation, using an algorithm called FeldKamp's algorithm. The latter integrates the geometric parameters of the scan in order to reconstitute tomographic images devoid of any artefact which could result, for example, from an alignment defect.
The images thus recomposed and colored can be used. The various dimensional measurements made from these are a reliable reflection of the subject. This technology reveals, at the finest level, the morphology of the entire jumping apparatus and makes it possible to model the various levers which interact within it, as shown in the following figure which shows the modeling of a furca in the unfolded position.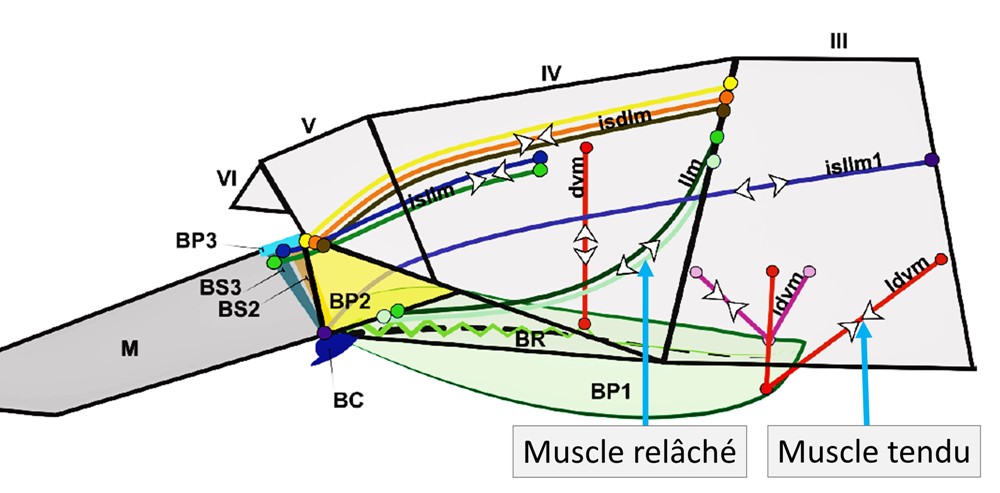
We note that each muscle, or bundle of muscles, is accompanied by pairs of arrows which, depending on their orientation, indicate whether the latter is tense or relaxed. This same model, established for other positions of the furca, indicates that the strategies and mechanisms involved during jumps vary between different species with distinct segmentations of cylindrical shape (Tomocerus.sp and Orchesella.sp) and that of globular shape. ( Sminthurus.sp, Bourletiella.sp) . The essential role of the basal plates (BP.) is, moreover, identified as the seat of the storage of the energy released in the form of kinetic energy at the time of the jump. The trigger function of the retinaculum, in addition to the already known one of locking the furca, is also confirmed.
Methodology of the study of the jump:
The modeling of the jumping system is an essential element in understanding how it works. However, it cannot free itself from a cinematic analysis . Indeed, the different phases of a jump: take-off, flight and landing, in addition to implementing the musculature described above, call upon other means of "steering" which springtails have recourse to. Below, diagram of the methodology implemented for the analysis of the flight of an Isotomurus retardatus which has an elongated and segmented body, comparable to that of Orchesella cincta .
Description : A sample of Isotomurus retardatus is placed on a shallow container covered with water (laboratory reconstruction of the environment in which this springtail evolves). This aquatic area is illuminated by a metal halide lamp producing an intense white light (neutral), essential for illuminating subjects during fast-paced shots.
 Springtail jumps are filmed at a rate of 1,000 to 10,000 im/sec. For the close-ups, we use a Canon MP-E65 mm lens (see chap. 35) and a Canon EF 200 mm lens for the wide shots. The small size of the springtails, the shortness of their flights and the limitation of the focal lengths of the optical assemblies used generate a lot of shooting waste.
Springtail jumps are filmed at a rate of 1,000 to 10,000 im/sec. For the close-ups, we use a Canon MP-E65 mm lens (see chap. 35) and a Canon EF 200 mm lens for the wide shots. The small size of the springtails, the shortness of their flights and the limitation of the focal lengths of the optical assemblies used generate a lot of shooting waste.
A long preliminary visualization work is necessary to extract exploitable sequences. In parallel with the technical aspect, linked to the production of the films, it is also necessary to ensure that certain measurements can be made, throughout the flight phases, such as the instantaneous speed υi or the angular position of the body ωi of the subject.
For this purpose, the researchers set up virtual landmarks on the modeled springtail body (illustration opposite).
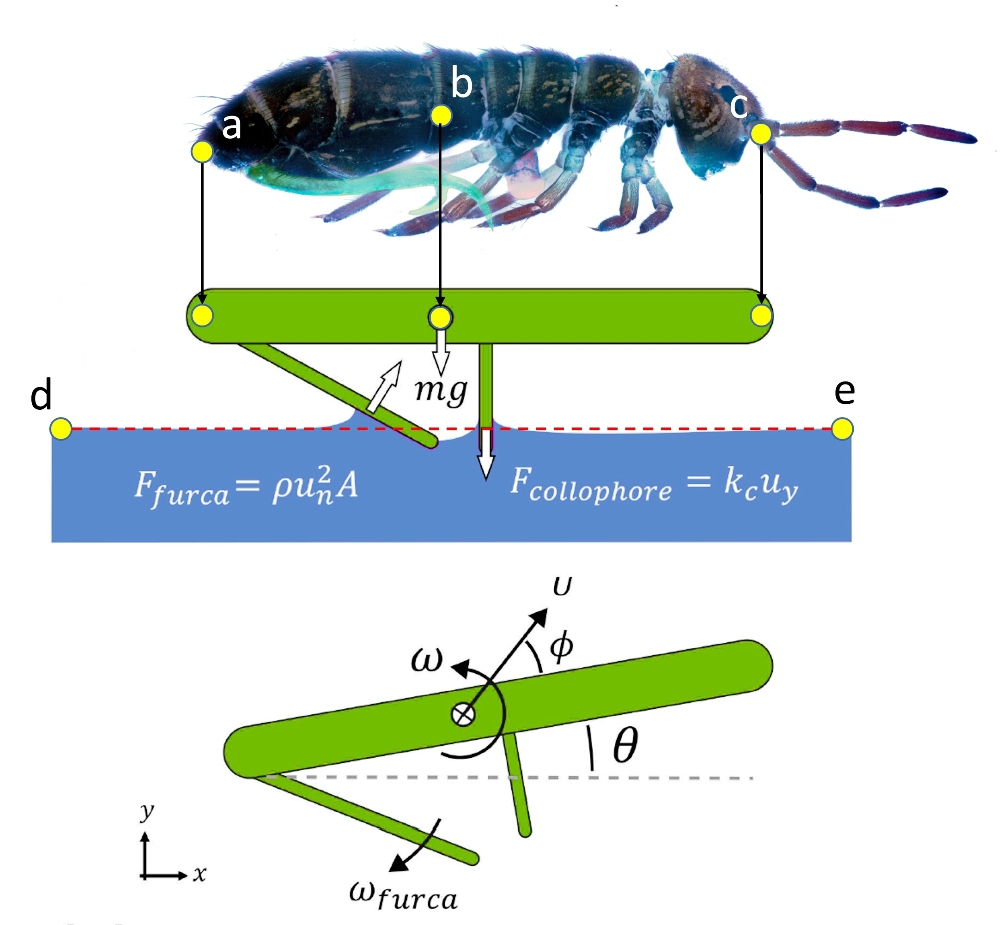 The digitization of three determinant points (Fig. on right - here in yellow) of the springtail, makes it possible to collect data throughout its trajectory according to a two-dimensional reference frame , relative to the central point (b) , considering é r é as the center of gravity. The two other landmarks, (a) and (c), respectively located at the end of the abdomen and at the tip of the head, make it possible to model the proper movements of the springtail which evolves on this trajectory.
The digitization of three determinant points (Fig. on right - here in yellow) of the springtail, makes it possible to collect data throughout its trajectory according to a two-dimensional reference frame , relative to the central point (b) , considering é r é as the center of gravity. The two other landmarks, (a) and (c), respectively located at the end of the abdomen and at the tip of the head, make it possible to model the proper movements of the springtail which evolves on this trajectory.
Finally, two distant points on the surface of the water, (d) and (e), establish the two-dimensional reference frame (x,y). Thus, thanks to these landmarks, we know, at each instant, the body posture, the take-off angle θ and the height relative to the water. From these values we deduce, by calculation, the instantaneous parameters of the flight, such as speed, acceleration, rotations, etc… These calculations being carried out on the basis of classical equations of kinematics or hydrodynamics.
The study that allows researchers to analyze different phases of the trajectory of the semi-aquatic springtail Isotomurus retardatus has shown that this species is able to perform directional jumps, rapid aerial recovery and landings, on the surface of the water, perfectly controlled. Below are eight views of a lift-off sequence on which the furca is colorized in yellow, the collophore in red and the water droplet in blue.
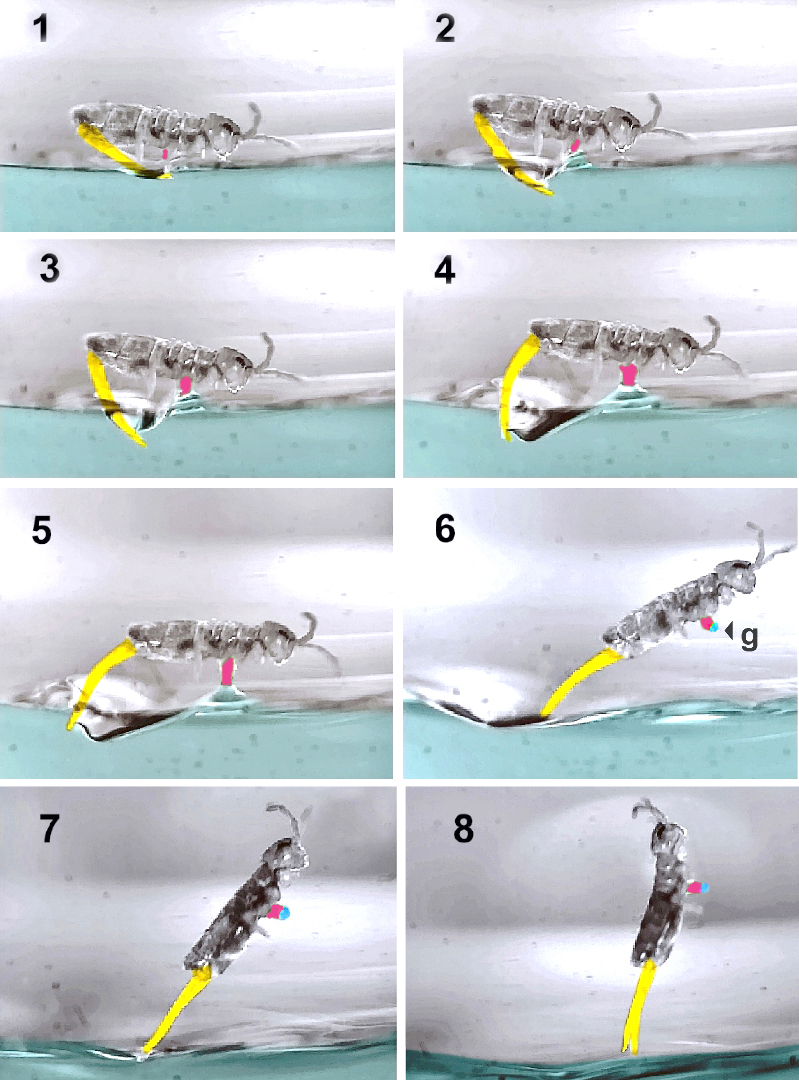
Description :
- Before jumping, the springtail anchors itself to the surface of the water using the collophore (1).
- At the beginning of the deployment of the furca, the latter remains in contact with the water (2, 3, 4, 5) and the tip of its abdomen is kept as far as possible from the surface.
- On take-off (6, 7, 8), the collophore traps a droplet (g) of water which weighs approximately 3% of the body mass and which acts as a counterweight, to help aerial recovery.
The time elapsed between each image is about 0.4 ms, which gives an idea of the lapse between the beginning of the relaxation and the moment when the furca is no longer in contact with the water, i.e. a total of 3.2 ms.
Below, modeling of a takeoff, which presents each angular position of the furca (manubrium = red, dente + mucron = green) with intervals of 0.2 ms. The very short durations measured during these events reveal impressive values: For example, the speed of movement observed reaches up to 280 times the length of the body per second. Isotomurus measuring about 2.3 mm, we deduce a speed during takeoff of 0.64 m/s, which, reduced to the scale of a man of 1.75m [1], corresponds to a supersonic speed of 1715 km/h (Mach 1.4). The measured accelerations are around 50 ± 30 m/s², i.e. between 2 and 5 G. For comparison, 2 G is the maximum acceleration of a formula one and 5G corresponds to the loss threshold of the central vision (blackout) of a fighter pilot.
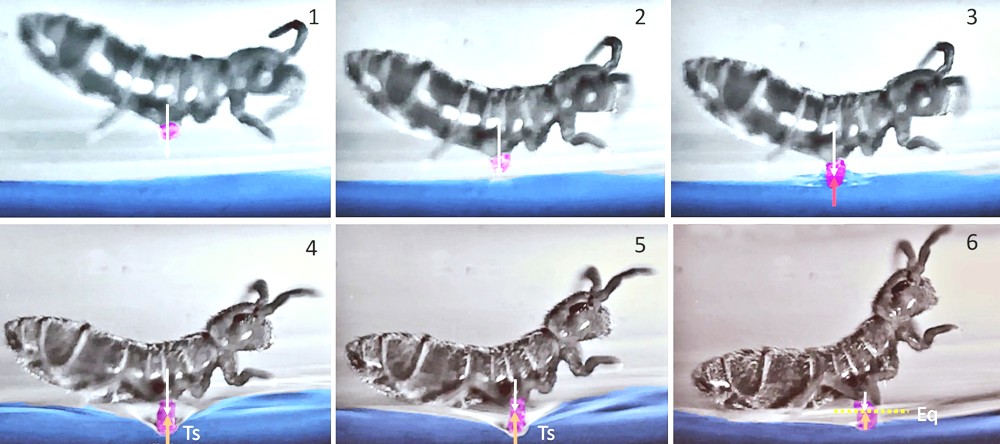
The calculations carried out from these models show that, during the initial phase of take-off, the force of adhesion to the water of the collophore enters, for a good part, into the directional control of the impulse. This adhesive force is due to the microstructure of the collophore and to the surface tension of the fluids (a property which allows springtails to remain on their feet on the surface of the water without sinking, or, to other animals, such as Gerris, to move on the surface of the ponds. The landings, also modeled, made it possible to establish the effectiveness of the collophore as a stabilizer on reception. On the next page, a succession of images shows the use of the collophore (coloured in red) during a reception.
After a jump, the springtail comes to the surface of the water (1, 2) with average impact speeds of 0.5 m/s. Due to the arcuate shape it adopts during its flight, contact with the liquid takes place via the collophore (3). It follows a passage below the water level, to a depth equivalent to 60% of the abdominal width, or 0.2 mm (4, 5). However, surface tension results in a curved deformation of the surface without the springtail being submerged. These same forces exert a counter-thrust (Ts) at the level of the contact zone (colophore/water) which makes it possible to reach the point of equilibrium (6). The springtail is then stabilized. Between the moment when the collophore comes into contact with water and the moment when stabilization is complete, approximately 4 ms. have elapsed here.
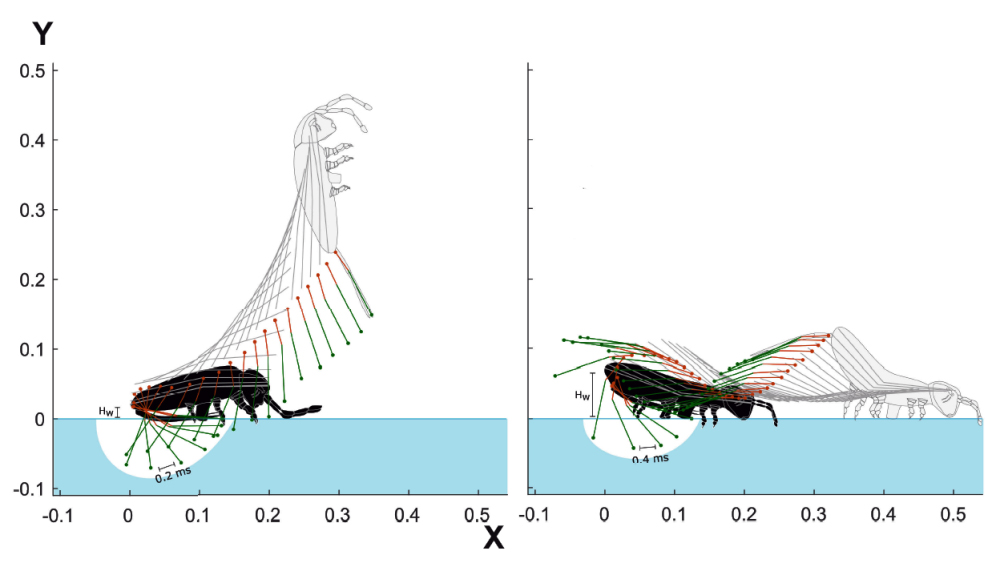
In complement to the evocation of the take-off and landing phases, let us now look at the flight phase, during which the springtail adopts three characteristic postures.
In addition to these three postures, mentioned on the following page, we note, when viewing the videos, that the springtail performs a large number of movements comparable to "contortions" which show that the latter operates, throughout its trajectory, low amplitude adjustments which could be intended to correct the latter or to perfect its positioning for reception.
In the diagram below, the trajectories of springtails are marked with colors:
- Trajectory of the head in red.
- Trajectory of the abdominal extremity in blue.
- Center point trajectory (approximate center of gravity) in black.
Description :
In the upward phase, the furca remains tense and the springtail performs a backward reversal on itself (counterclockwise). During the flight, it folds up against the abdomen and the springtail can then perform a variable number of rotations. At the beginning of the descending phase, the springtail adopts an arched posture, conducive to landing.
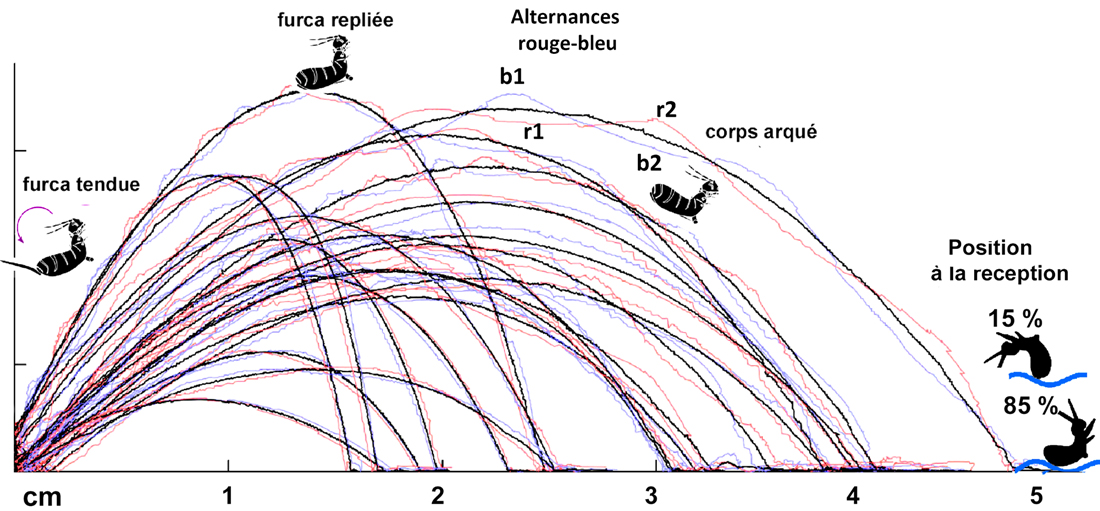
the aerial part, under the zone marked "red -blue alternation" I spotted two portions of trajectory [b1- b2 ] and [r1-r2] . It can be seen that the latter alternate their position (above/below) with respect to the black trajectory. Each alternation corresponds to a rotation of the collembola on itself and makes it possible to count the total number of rotations carried out during the flight.
85% of springtails, when they arrive, are in an arched posture, which allows the collophore to be the point of first contact with water. This reception mode induces almost perfect landings. 15% of landings are made, on the other hand, in positions that do not allow such rapid stabilization. In these cases, the springtails that come into contact with the water in less appropriate postures receive each other randomly on various parts of their body. The collophore cannot then play its role.
However, the stabilization will occur following recovery movements generally carried out during the rebound(s). The shots then reveal fairly eventful recoveries, as illustrated in the figures below.

Description :
- The images (1, 2) show a landing in an inverted position (on the back).
- The antennae touch the water first and the springtail crashes to the surface (3, 4).
- It initiates a rebound (5).
- Turning over on itself (6, 7).
- Landing on the head, but in ventral orientation (8).
- End of turning allowing the collophore to come into contact with water (9).
- The collophore plays its role of stabilizer, aided by the extended hind legs (10).
The landing phases which were timed showed that a stabilization carried out from a bad landing position can last up to 10 times longer than a ventral type landing with immediate use of the collophore, i.e. approximately 44 ms.
There are some positions adopted as needed during a flight:
 - Straightening in an arched position (1).
- Straightening in an arched position (1).
- Damping rotation in arched position with outstretched legs (2).
- Fold in the shape of a ball, legs against the abdomen, to accelerate the descent (3)
- Upside down posture of a dead Springtail (4)
- Aerodynamic posture with tense furca at the start of flight (5)
- Use of forelegs for cushioning on landing (6)
In springtails with a globular morphology, Bourletiella hortensis, mastery of jumps seems less established. A study [5] carried out by researchers from the Japanese University of Tohuku shows that the latter performs jumps whose profiles are distinct from those of Arthropleones.
The conditions of the experiment differ from those previously described due to the nature of the substrate. Indeed, Bourletiella hortensis evolves on solid grounds which exert, on the supports of the furca, an incidence different from that of a liquid support.
Another important determinant is the globular shape of the springtail whose aerodynamic profile differs significantly from that of a springtail with a cylindrical body. The researchers observed different types of trajectories which show, for the first (fig.A below) , an upward phase close to the vertical (angle with the ground ≈ 86°) and a substantially symmetrical descent.
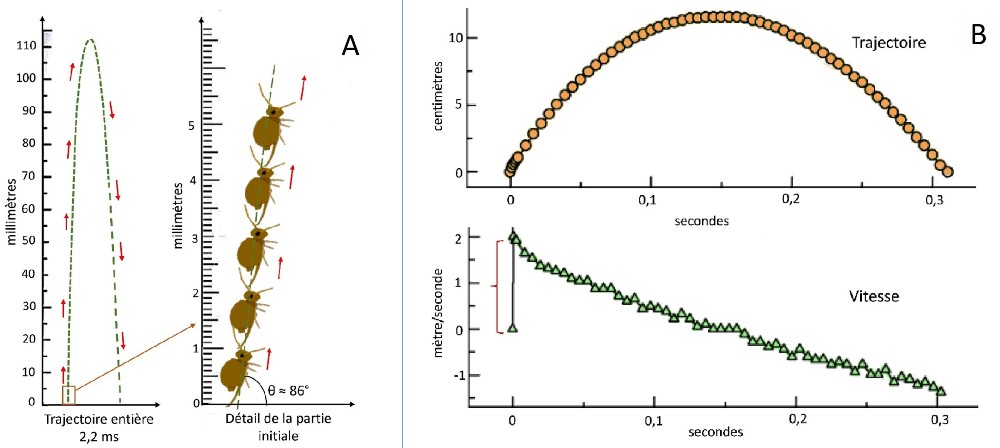
The distance traveled here is small. It allows the springtail to flee a predator, but cannot really be considered as an ordinary means of locomotion. This springtail can however perform longer jumps, as shown Fig.B above, below where the distance traveled is equal to approx. 100 times body size.
This value must be modulated by the fact that a globular collembola performs during its flight up to 417 rotations/second. Reduced to the average duration of flight (0.2 sec.), we obtain 83 rotations, that is to say about 10 times more than that generally observed during the jumps of Arthropleones. The maximum height reached is comparable to that recorded on the previous trajectory, i.e. 11.5 cm.
The maximum take-off speed very quickly reaches 2 m/s, then it decreases linearly.
The acceleration reaches 300 m/s² in 1.65 ms., during the phase marked with a red brace.
Videos acces :
The different phases of the flight of isotomurus retardatus are difficult to translate with photographic montages. Below is access to excerpts of videos from which the montages of this article are taken :
- Isotomurus retardatus taking off
- Isotomurus retardatus landing
- Isotomurus retardatus flight postures
Conclusion :
The semi-aquatic springtail partly controls its trajectory. His movements during the flight, allow him in 85% of the cases to adopt the proper posture for an optimal reception.
It would be useful to know to what extent the other postures adopted by the springtail during flight allow it to act on parameters such as its Cx (drag coefficient) or its lift. We could also study the behavior of other springtails with comparable morphologies evolving on different substrates to look for similarities or specificities during flights.
Studies of the jumps of Symphypleones reveal less controlled trajectories and more random landings.
Sources :
[1]: Victor M. Ortega-Jimenez a,b, Elio J. Challitaa,c, Baekgyeom Kimd, Hungtang Koc , Minseok Gwond, Je-Sung Kohd, and M. Saad Bhamlaa
aSchool of Chemical and Biomolecular Engineering, Georgia Institute of Technology, Atlanta, GA, USA; bSchool of Biology and Ecology, University of Maine, ME, USA;
cGeorge W. Woodruff School of Mechanical Engineering, Georgia Institute of Technology, Atlanta, GA, USA; dDept. Mechanical Engineering, Ajou University, Republic of Korea
[2]: Godrie Baptiste, “Egalitarian relationships in the production of scientific knowledge. The example of participatory research in mental health", Vie sociale, 2017/4 (n° 20), p. 99-116. DOI: 10.3917/vsoc.174.0099. URL: https://www.cairn.info/revue-vie-sociale-2017-4-page-99.htm
[3]: “On springtails (Hexapoda: Collembola): a morphofunctional study of the jumping apparatus”
Fábio Gonçalves de Lima Oliveira* - Frontiers in Zoology (2022)/ open access.
[4]: This type of comparison is only valid to illustrate my point, because the impact of accelerations on structures such as a human body, do not respond, relative to their mass, to linear values because they involve cubic factors .