1) Introduction
While some predatory insects hunt "on the run" or in flight, others adopt a motionless position and capture their prey at a distance with their front legs or their modified mouthparts. This is the case for mantis, mantis religiosa (1) or certain bugs Empicoris rubrumaculatus (2) which project their ravishing forelegs or dragonfly larvae (3) with their lower lip transformed into a prehensile organ (called the mask).
A quick attack coupled with excellent visual acuity often ensures successful capture of coveted game.
This article draws on the work of several researchers (references at the end of this work) and highlights two predation strategies used by all species of a genus of Staphylinidae, the Stenus Latreille, 1796. These staphylines use, in the adult state, either their mandibles, or a "harpoon" to catch their prey, often springtails.
To illustrate the following article, click on the image below to access a video which shows rove beetle spearing.
 https://www.youtube.com/watch?v=RjKhIg3kCpM
https://www.youtube.com/watch?v=RjKhIg3kCpM
2) The predator

A Stenus is a beetle with an extensible labium (complex lower lip), which allows it to practice stalking or hunting. Its prey moves, like it, on the ground or on plants. This is the case of certain species of springtails which, in addition to escape by running away, can also jump using their furca (jumping organ which is triggered at the slightest stimulus).
This escape takes place at substantially identical speeds in different species of springtails; and therefore, if the Stenus wants to catch these springtails, it will have to, at least, run as fast.
The labium of the Stenus is able to lengthen at lightning speed to reach its prey and immobilize it thanks to special "pads", as we will discover. The labium then rapidly retracts and the prey is seized by the two thin mandibles.
As part of a study conducted on the hunting practiced by the Stenus, German entomologists observed three species of springtails (the prey) and also noted that, for almost half of its attacks, the Stenus use their extensible labium .
Photo opposite: Stenus bimaculatus Gyllenhal, 1810 © A.Wojtyra.
3) The preys
The photo-montages below illustrate the morphologies of the three species of springtails that served as prey for the Stenus. Inset, detail of their cuticular coatings which will be decisive in the success of the captures.
Heteromurus nitidus (Templeton in Templeton & Westwood, 1836): Entomobryomorph that moves on the surface on rather dry soils, its body is covered with scales and a few long bristles.

Heteromurus nitidus: photograph of an individual in dorsal view in its natural biotope © Ph.Garcelon; inset, scanning electron microscope photo of the abdomen showing the scales ,© T.Bauer & M.Pfeiffer.
Podura aquatica (Linné, 1758): Poduromorph that likes wetlands and frequently moves on the surface of the water. Its body is covered in small tubercles with very few short bristles.
 Podura aquatica: photograph of an individual in dorsal view in its natural biotope © Ph.Garcelon; inset, scanning electron microscope photo of the abdomen , © T.Bauer & M.Pfeiffer, showing the microtubers and the few sensory bristles.
Podura aquatica: photograph of an individual in dorsal view in its natural biotope © Ph.Garcelon; inset, scanning electron microscope photo of the abdomen , © T.Bauer & M.Pfeiffer, showing the microtubers and the few sensory bristles.
Isotomurus palustris (Muller, 1776): Entomobryomorph which mainly evolves in humid areas immediately bordering ponds or lakes. Its body is entirely covered with bristles.
 Isotomurus palustris : photograph of an individual in dorsal view in its natural biotope © Ph.Garcelon; inset, scanning electron microscope photo of the abdomen, © T.Bauer & M.Pfeiffer, showing the different sensory bristles.
Isotomurus palustris : photograph of an individual in dorsal view in its natural biotope © Ph.Garcelon; inset, scanning electron microscope photo of the abdomen, © T.Bauer & M.Pfeiffer, showing the different sensory bristles.
4) Let's find out about these predatory Stenus
Let's leave our springtails for a moment without jumping to conclusions, and let's look in more detail at the Stenus, its morphology and its "armament".
The Steninae subfamily includes more than 3300 species distributed between two present-day genera1: the Stenus Latreille, 1796 and the Dianous Leach, 1819. 24 fossil species have also been described in these two genera, but also in other genera such as Eocenostenus Cai, 2014 (one species from the Paleogene2 from France) and Festtenus Zyla, 2017 (2 species from the Upper Cretaceous3 from Burma). The Steninae are beetles belonging to the very diverse Staphylinidae family, one of the taxa, with the Curculionidae (or weevils), the richest in species of the animal kingdom (more than 65,000 known species).
Just over 3000 species appear in the genus Stenus alone, making it the second largest on an animal scale. The vast majority of species live in tropical forests. A little less than 150 species are observed throughout France.
Their distribution is worldwide, with the exception of Antarctica and New Zealand.
These staphylines, small to medium sized (± 1 to 10 mm) are most often observed: (1) in habitats associated with moist humus or plant debris near the ground, (2) on bare ground, or (3) on plants frequently at the water's edge.
The Stenus are vagile species, therefore pursuing their preys on the ground or among the vegetation, but never while flying. They hunt many invertebrates on sight, such as oligochaete worms (such as earthworms), mites, spiders, aphids or even (and especially) springtails. Like other predators, they capture their prey with their two scythe-like mandibles. Or, they catch their "game" on the lookout and from a distance thanks to a unique "telescopic" labium, used like a harpoon. It is one of the most specialized prey capture structures in insects. We will come back later.
This sub-family is easily recognized by their very typical general habitus.
- 1 A third Australian genus awaits description.
- 2 Upper Cretaceous: Geological period between 100.5 and 66.0 million years ago. It ends with a massive extinction, the Cretaceous-Tertiary extinction.
- 3 Paleogene: geological period succeeding the Cretaceous which extends from 66.0 to 23.03 million years ago.
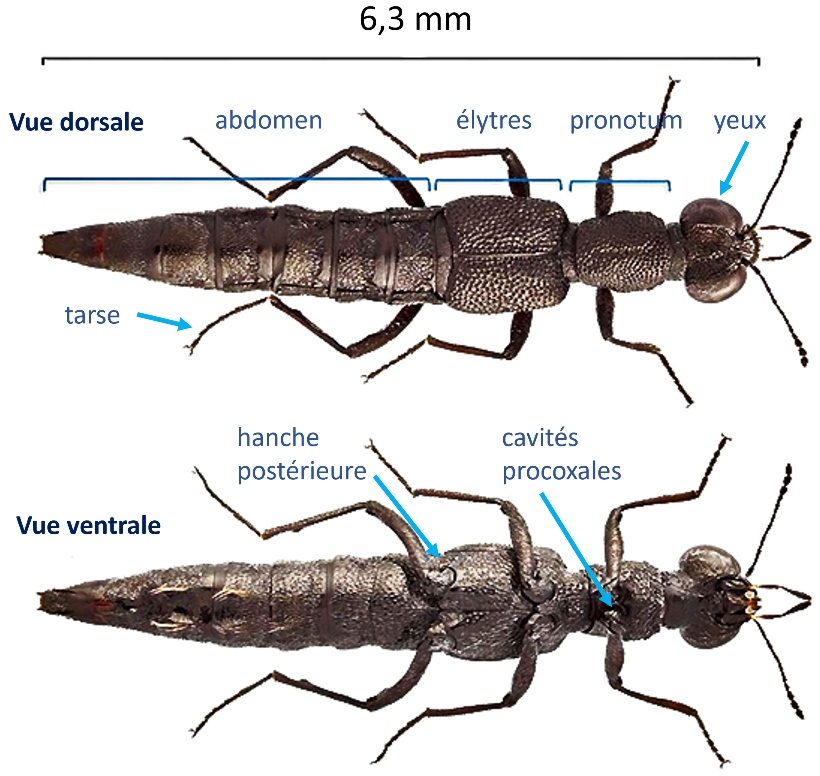 Habitus of Stenus ater - Mannerheim, 1830. Modified from a photo by © E. Gay.
Habitus of Stenus ater - Mannerheim, 1830. Modified from a photo by © E. Gay.
Adult Steninae (see photos above) have two large hemispherical eyes that occupy almost the entire sides of the head, from the base of the mandibles to the neck. They have very many ommatidia evenly distributed over the entire ocular surface, which explains the very good visual acuity of these staphylines.
The antennae consit of 11 segments or antennomers (with the two thicker basal antennomers and a more or less marked terminal club composed of 3 antennomers); they originate on the vertex between the eyes.
The pronotum – dorsal part of the prothorax – has a cylindrical shape without lateral margins. The short elytra are lined with a dense piligerous punctuation, they do not protrude from the metathorax and do not have a lateral (epipleural) carina.
The anterior hips are poorly developed; conical in shape, they lodge and articulate in completely closed anterior coxal cavities. The posterior hips are clearly separated. All tarsi have five articles (pentamers).
The abdomen is elongated and has a cylindrical section, a rim is present or not on the tergites. The aedeagus (genital structure of the male) is symmetrical (photos below).
 Aedeaeus of Stenus ater. Maximum length: 0.7 mm - modified after © E. Gay.
Aedeaeus of Stenus ater. Maximum length: 0.7 mm - modified after © E. Gay.
This organ includes a central part, the median lobe, on which two paramers are articulated. The spermatheca of females is sclerotized or not. The Dianous are easily distinguished from the Stenus by two more or less long styles at the end of the abdomen.
Let us return and detail this "harpoon", characteristic of the Stenus.
While the labium of Dianous beetles is short and unspecialized, Stenus species are all characterized by an elongated, prominent labium that functions as a prey capture device at a distance. Note that it was already present in fossil species of the genus Festtenus.
The labium can be projected towards a potential prey in a few milliseconds and this, by the only pressure of the hemolymph. Once touched by the adhesive pads, the prey is fixed and, after rapid retraction of the labium (30-40 m/s), grasped by the beetle's mandibles. Note that all the Stenus studied so far are also able to catch their prey directly with the mandibles without using their specialized labium.
The adhesive labium of Stenus is particularly effective, and makes these staphylines very formidable predators. It can attach to both smooth cuticles and cuticles with varied microsculptures present in a wide range of potential prey (see photo of springtail cuticles above or click here).
The "labial harpoon" is composed (photo below) of an elongated rod-shaped distal prementum connected to the head by an equally stretched conjunctiva membrane. At rest, it is invaginated in the head like an upturned finger inside a glove. The capture device works like a catapult: before the strike, the labium is compressed by the pressure of the haemolymph, then is suddenly released, and thrown forwards from the body in 4 to 5 milliseconds.
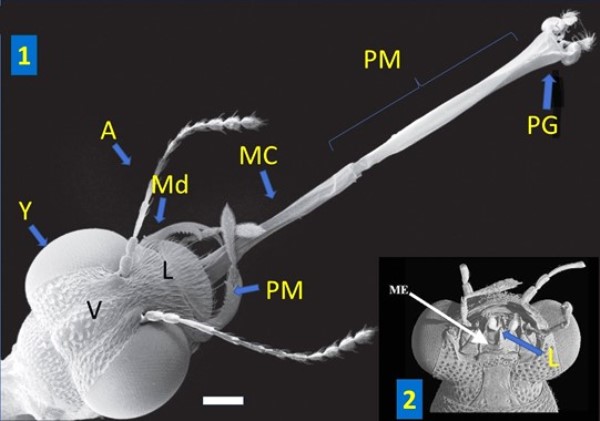 Scanning electron microscope photos of heads of Stenus sp.
Scanning electron microscope photos of heads of Stenus sp.
1. Stenus bimaculatus Gyllenhal, 1810 with the labium devaginated. Dorsal view. A: antenna, Md: mandible, PM: maxillary palp, L: labrum, MC: connective membrane, ME: mentum; PG: paraglossus, PM: prementum, V: vertex, Y: eyes. Modified from © Betz, 2016. Scale: 200 µm.
2. Ventral view of the head of Stenus sp. with the labium (L) in the rest position. Modified from © Żyła et al., 2017. Scale: 200 µm.
At its end, the prementum (see photo below) has two paraglosses modified into adhesive pads made up of a multitude of branched trichomes (long hollow bristles) which increase the adhesive surface.
The shape of these paraglosses (and therefore the adhesive surface) has evolved according to the prey sought by the various species of Stenus. Thus, there are ellipsoidal, oval, elongated, conical pads...
The connecting membrane and the prementum are traversed by bundles of longitudinal canaliculi which transport, towards the adhesive pads, the adhesive secretion produced by secretory glands located in the head. The latter are made up of separate glandular units, each composed of three cells. Two of these cells are active secretory cells, one producing protein secretions, the other lipoid secretions. Consequently, a two-phase secretion is observed on the paraglossus. The glandular canaliculi which go from the glands to the paraglossus also serve as a reservoir for secretions before their evacuation just before the attack of a prey.
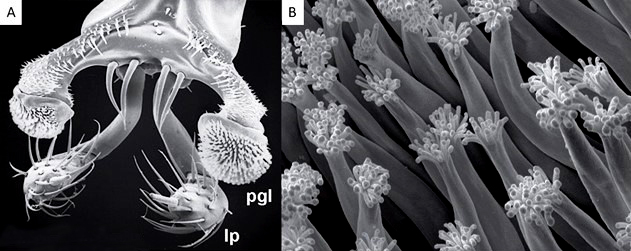 Scanning electron microscope photos of heads of Stenus sp.
Scanning electron microscope photos of heads of Stenus sp.
A. Stenus comma LeConte, 1863. Vue dorso-frontale de l’extrémité du labium dévaginé. Pgl : paraglosse et lp palpe labial. Échelle 100 µm. Modifié d’après © Betz, 2006.
B. Stenus bimaculatus Gyllenhal, 1810. Détail de l’extrémité des trichomes adhésifs. Échelle 2 µm. Modifié d’après © Koerner et al., 2016.
Van der Waals forces1 as well as surface tension2 and the viscosity of adhesive secretions are considered as possible mechanisms of the adhesion necessary to capture springtails, for example.
The sticky gel is invaded by a large number of bacteria belonging to Actinomycetales (Arthrobacter, Microbacterium, Rhodococcus) and Pseudomonadales (Acinetobacter, Pseudomonas). Their function remains to be defined. They may increase the adhesive properties of the gel lining the pads.
The presence of resilin in the wall of the trichomes makes them very flexible and elastic, so that they can adapt tightly to the shape and surface irregularities of the prey (see article on this site: microstructure of the cuticle of springtails ). As the "labium-harpoon" is used several hundred times during the life of the beetle to capture prey, resilin provides the resistance of the pads to wear, similar to the function of resilin in the folds of the staphylines wings, folded - at rest - many times under the elytra.
Compared to the possibility of capturing an insect by simply grasping it with the mandibles, the adaptive value of the "harpoon-labial" lies both in its superior execution speed and reach, as well as in its ability to fix the prey. at the time of contact.
Thus, even Stenus with limited activity and reactivity are able to capture otherwise elusive invertebrates surprisingly quickly and efficiently.
Nevertheless, the technique of capturing prey by the mandibles has also evolved in certain very agile and reactive Stenus, living on the ground in open habitats: the largest prey - with powerful flight movements - are elusive with the "labium-harpoon », the adhesion forces of the pads not being sufficient to hold them firmly, they are then caught and immobilized with the mandibles. This immobilization is reinforced by the large tooth and the denticles dotting the internal side of the fine mandibles.
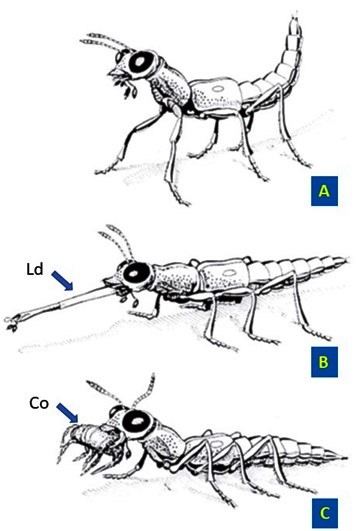
The drawings opposite illustrate the three phases of a “harpoon” hunt by Stenus comma LeConte, 1863.
(Insect size: 5.5 mm. Modified from © Betz, 2006).
A. The Stenus visually located its prey and positioned itself at a distance allowing it to reach it.
B. By exerting strong pressure on its haemolymph, it devaginates its labium (Ld) which propels itself at very high speed towards its prey.
C. Once its prey (Co) has been seized by the adhesive trichomes that cover its paraglosses, the labium retracts and brings the prey back towards the mandibles.
We now know the Stenus better, and can come back in more detail to its attack strategies, in this case against the three species of springtails mentioned above.
- 1 Corresponds to a weak electrical interaction taking place at short distances between atoms and/or molecules.
- 2 Can be briefly defined as the force that exists at an interface between two media.
5) Strategies: Attack-Defense
A springtail detects the approach of a predator by several sensory pathways such as the eyes or thanks to mechano-sensory and/or chemical perceivers (see articles on this site devoted to the sensory organs: Silks and sensilla and PAO )
Generally, large springtails avoid direct contact by fleeing forward, a fairly constant distance. This escape behavior already operates a selection among predators and discards the most clumsy or slowest species of Stenus.
How did the Stenus, during their co-evolution with springtails, optimize their hunting?
The extendable labium, used like a harpoon, allows a greater predator/prey distance which will less arouse the vigilance of the prey. The researchers also observed that during their attacks, the "harpooning" Stenus rush towards their prey simultaneously with the deployment of their labium, this movement reduces the probability of springtail escape and increases the probability of capture.
The running speed measured on samples of "large" springtails (average size adult stage: 2.5 to 3.0 mm) is between 1.77 and 2.78 mm/s and it is between 0.90 and 1.18 mm/s for the small ones (size average adult stage ≤ 2.0 mm) These speeds explain in particular why large springtails are more difficult to capture by Stenus. As already mentioned, the body mass of springtails is also an important factor to correlate with the adhesive capacity of the predator's labium. The heavier the prey, the greater the adhesiveness of the end of the labium.
Finally, certain Stenus (such as S. comma LeConte, 1863 - S. fossulatus Erichson, 1840 and S. canaliculatus Gyllenhal, 1827) which hunt in open ground, have refined their predation techniques to the point of being able to seize large springtails using their mandibles with a high success rate (25 to 40%). Conversely, the success rate is only 2% for attacks by S.fossulatus using its labium to reach a "large" springtail.
Without going into the details of the various studies conducted on the subject, it can be argued that, in general, Stenus rather hunt small springtails using their labium, while they tend to use their mandibles to capture large springtails.
6) Behavioral study of springtails
Data and movement counts for three springtail species are summarized in the table below. They were measured on 5 individuals of each species. The specimens studied were placed in a glass cuvette and filmed for 11 minutes. As these results indicate, the behaviors of these taxa are not homogeneous and may even show strong variations.
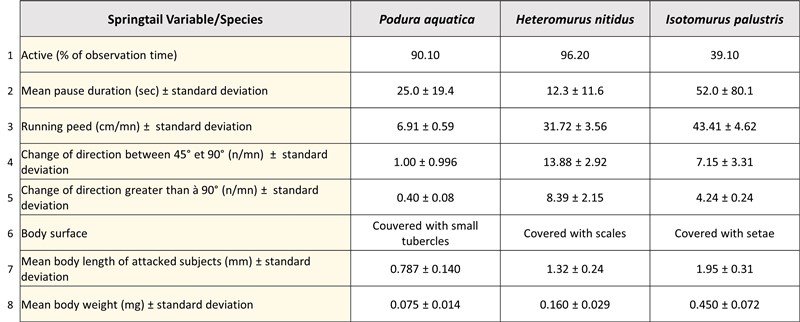
Table analysis :
- Line 1: average percentage of springtails active during the observation time. P. aquatica and H. nitidus stand out clearly and are more than twice as active as I. palustris. Note that one of us (Ph. G.) observed that H. nitidus, living on the superficial layers of the soil and on firm ground, moves with few pauses and rummaging in all directions. The P. aquatica, on the other hand, move quite slowly and, as they often evolve in aquatic areas, they are very agitated, without their movements being very efficient, and this, because of the pointed claws of the extremity. of their legs, poorly adapted to moving on the surface of the water (the lift being provided by the surface tension force of the water).
- Line 2: average duration of the breaks made by the 3 species of springtails between their active periods. I. palustris, with its pauses two to four times longer than the other two species, would be an easier target for a stalking predator like a Stenus.
- Line 3: escape velocity per stroke in cm per minute. I. palustris is significantly faster than P. aquatica and, to a lesser extent, than H. nitidus. Based on this parameter, P. aquatica has a greater risk of being “harpooned” by a Stenus.
- Lines 4 and 5: number of trajectory changes per minute with an angle respectively between 45° and 90° and greater than 90°. These lines indicate that P. aquatica moves almost straight, while H. nitidus changes course on average every 3 seconds and I. palustris every 5 to 6 seconds, which confirms the observations of one of between us (Ph.G.). Therefore, based on these trajectories, P. aquatica would be easier prey for a Stenus.
- Line 6: The adhesion of the pads of the end of the labium (see text and photos above for more details) will be all the more important and strong on an uneven surface rather than on a smoother surface, as in P. aquatica (see photos below).
Rows 7 and 8: average size and weight of springtails. The larger and heavier the species (I. palustris), the more chance it will have of escaping the "harpoon" of the Stenus.
All these rather disparate data underline that each of the three springtail species does not have the same defensive capacities. A priori, faced with a predator like a Stenus, P. aquatica would seem to be relatively disadvantaged in relation to its slow movement speed, its very low ability to change direction, its smaller size and weight, but also its ecology ( it evolves in habitats that the Stenus particularly like).
 Podura aquatica on the water, on the edge of a pond at an altitude of 1800 m © Ph. Garcelon.
Podura aquatica on the water, on the edge of a pond at an altitude of 1800 m © Ph. Garcelon.
7) Analysis of attacks on springtails
Prerequisite: Observation and counting of attacks require the installation of elaborate camera and lighting equipment that we will not detail here. It should be noted, however, that to film these very rapid attack movements (measurable in milliseconds), the researchers used cameras capable of recording images at rates of 1000 to 1050 frames/second.
To make their observations, springtails were placed in identical situations facing their predators. The Stenus were taken from their natural habitat, on the edge of a sandpit pond, then stored in boxes, on a moistened plaster of Paris substrate, maintained at a constant temperature of 20°C. They were voluntarily starved for four to five days. The springtails were captured in the habitat of the Stenus or come from cultures carried out in the laboratory. Also kept on damp plaster but fed with soy flakes.
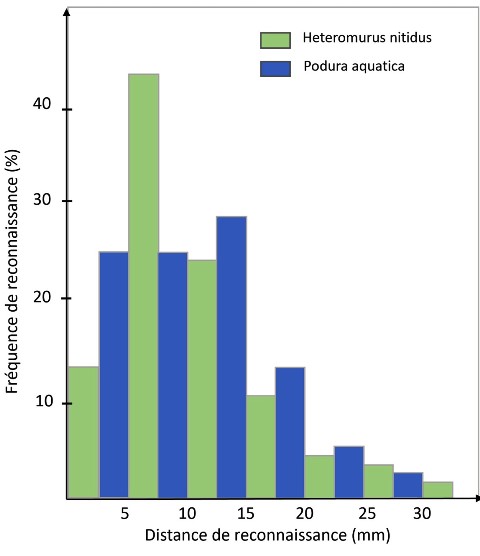
The researchers first tried to assess the distance at which the Stenus spotted its prey. For this, they chose to confront the Stenus with Heteromurus nitudus (1.5 mm) and Podura aquatica (0.8 mm) because of their significant difference in size. For their experiment, they used boxes 6 cm in diameter in which they, each time, confronted a Stenus with three springtails of the same species. They used 18 Stenus to carry out this experiment, at the end of which they were able to evaluate 160 events per springtail species.
The histogram opposite summarizes the distributions of the recognition frequency in relation to the distance between the prey and its predator.
The recognition distance being defined as that between the end of the staphylines head and the part closest to its prey.
The table below indicates that the recognition frequency reaches its maximum (+40 %) for Heteromurus nitidus between 6 and 10 mm. Beyond 15 mm, it drops to less than 10 %. For Podura aquatica, the amplitudes as a function of distance are less accentuated and the range wider (between 5 and 20 mm, with an average recognition rate slightly above 25 %).
More generally, we note that beyond 15 mm the recognition rate is close to 10 % and drops around 5% beyond 20mm to drop to less than 1 % for distances greater than 30 mm.
8) Discussion
The situations reproduced by the researchers are not quite identical to those encountered by the populations studied within their respective habitats. Indeed, the nature of the areas in which they evolve is difficult to translate into figures because too many variables have to be taken into account. However, the figures given here represent the best possible approach in the laboratory. Ideally, the measurements should be taken "in situ", which in practice is extremely difficult to achieve.
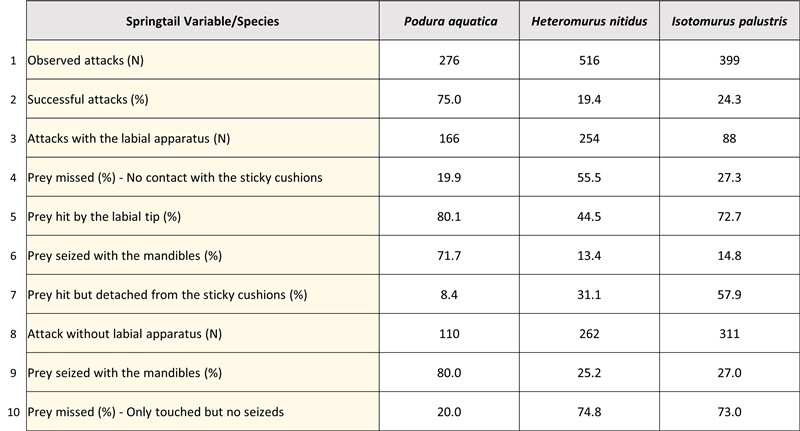
The Stenus attack analysis table reports a lower success rate (row 2) with larger springtails. Indeed, in line 8, we note that the large springtails (H. nitudus 1.32 mm and I. palustris 1.95 mm), covered with scales (H. nitidus) or bristles (I. palustris) show a greater propensity to detach from the end of the labium (line 7). It should also be noted that P. aquatica, whose trajectory is the straightest, and has the lowest escape speed (6.9 cm/min) (H. nitidus: 31.7 cm/min and I. palustris: 43.4 cm/min ) is the most exposed species (line 10).
Finally, if we calculate the percentage of attacks carried out with the mandibles (without using the labium) compared to the total number of attacks, we find 39.8% for P. aquatica, 58.8% for H. nitidus and 77.9% for I. palustris.
This would tend to show that Stenus are able to integrate certain variables relating to their prey (size, flight speed, change of direction, etc.) to make their strategic choices and best adapt their hunting mode.
Bibliography :
- Bauer TH. & M. Pfeiffer (1991). 'Shooting' springtails with a sticky rod: the flexible hunting behaviour of Stenus comma (Coleoptera; Staphylinidae) and the counter-strategies of its prey. Anim. Behav., 41 : 819-828.
- Betz O. (1996). Function and evolution of the adhesion-capture apparatus of Stenus species (Coleoptera, Staphylinidae). Zoomorphology 116:15–34.
- Betz O (1998). Comparative studies on the predatory behaviour of Stenus spp. (Coleoptera: Staphylinidae): the significance of its specialized labial apparatus J. Zool., Lond. 244, 527-544.
- Betz, O (2006). Der Anpassungswert morphologischer Strukturen: Integration von Form, Funktion und Ökologie am Beispiel der Kurzflügelkäfergattung Stenus (Coleoptera, Staphylinidae). Entomologie heute 18 : 3-26.
- Betz O., Koerner L. & L. Dettner (2018). The Biology of Steninae. In: Betz O., Irmler U. & J. Klimaszewski J. (eds) : Biology of Rove Beetles (Staphylinidae). Springer, Cham. 229-283. https://doi.org/10.1007/978-3-319-70257-5_11.
- Cai C., Clarke D.J. Yin Z., Fu Y. & D. Huang (2019). A specialized prey-capture apparatus in mid-Cretaceous rove beetles. Current Biology, Vol. 29 (4) : R116 - R117. https://doi.org/10.1016/j.cub.2019.01.002
- Koerner L., Stanislav N., Gorbb S.N. & O. Betz (2011). Functional morphology and adhesive performance of the stick-capture apparatus of the rove beetles Stenus spp. (Coleoptera, Staphylinidae). Zoology Vol. (115, 2) : 117-127. https://doi.org/10.1016/j.zool.2011.09.006.
- Koerner L., Gorb S. & O Betz (2012). Adhesive performance of the stick-capture apparatus of rove beetles of the genus Stenus (Coleoptera, Staphylinidae) toward various surfaces. Journal of Insect Physiology, vol. 58 (1) : 155-163. https://doi.org/10.1016/j.jinsphys.2011.11.001.
- Koerner L. , Laumann M., Betz O. & M. Heethoff (2013). Loss of the sticky harpoon – COI sequences indicate paraphyly of Stenus with respect to Dianous (Staphylinidae, Steninae), Zoologischer Anzeiger - A Journal of Comparative Zoology, Vol. 252 (3) : 337-347. https://doi.org/10.1016/j.jcz.2012.09.002.
- Koerner L., Braun V. & O. Betz (2016): The labial adhesive pads of rove beetles of the genus Stenus (Coleoptera: Staphylinidae) as carriers of bacteria. Entomologia Generalis, 36 (1) : 33-41. https://doi.org/10.1127/entomologia/2016/0308.
- Koerner L., Garamszegi L.Z. , Heethoff M. & O. Betz (2017). Divergent morphologies of adhesive predatory mouthparts of Stenus species (Coleoptera: Staphylinidae) explain differences in adhesive performance and resulting prey-capture success. Zoological Journal of the Linnean Society, 181, 500–518.
- Kölsch G. (2000). The ultrastructure of glands and the production and function of the secretion in the adhesive capture apparatus of Stenus species (Coleoptera: Staphylinidae). Can. J. Zool. 78: 465–475.
- Żyła D., Yamamoto S., Schwenninger, W. K. & A. Solodovnikov (2017). Cretaceous origin of the unique prey-capture apparatus in mega-diverse genus: stem lineage of Steninae rove beetles discovered in Burmese amber. Sci Rep 7, 45904 (2017). https://doi.org/10.1038/srep45904.